-
 Lewandowski to leave Barca with 'mission complete'
Lewandowski to leave Barca with 'mission complete'
-
Pope Leo to visit France September 25-28

-
 Trump, Nigeria claim killing of senior IS leader
Trump, Nigeria claim killing of senior IS leader
-
Acosta takes pole, Bezzecchi crashes in Catalan MotoGP qualifying

-
 Arbeloa 'happy' if Mourinho back at Real Madrid next season
Arbeloa 'happy' if Mourinho back at Real Madrid next season
-
Fiery Finns, Australian star favourites at boycotted Eurovision final

-
 Haaland to play marauding Viking in new animated film
Haaland to play marauding Viking in new animated film
-
Lyles excited to race 'good kid' Gout over 150m

-
 'Parasite' director Bong says making animated film to 'surpass' Miyazaki
'Parasite' director Bong says making animated film to 'surpass' Miyazaki
-
World Cup fever gets tail-wagging twist as Singapore kits out pets

-
 France-born Bouaddi approved to play for Morocco before World Cup
France-born Bouaddi approved to play for Morocco before World Cup
-
South Korea coach backs Son to shine at his fourth World Cup

-
 Putin to visit China May 19-20, days after Trump trip
Putin to visit China May 19-20, days after Trump trip
-
Eurovision gears up for boycotted final, with fiery Finns favourites

-
 Son Heung-min to lead South Korea squad at his fourth World Cup
Son Heung-min to lead South Korea squad at his fourth World Cup
-
Pretty in pink: Dallas World Cup venue chasing perfect pitch

-
 Wordle heads to primetime as media seek puzzle reinvention
Wordle heads to primetime as media seek puzzle reinvention
-
Eurovision: the grand final running order

-
 McIlroy, back in PGA hunt, blames bad setup for lead logjam
McIlroy, back in PGA hunt, blames bad setup for lead logjam
-
Kubo vows to lead Japan at World Cup with Mitoma out

-
 McNealy and Smalley share PGA lead at difficult Aronimink
McNealy and Smalley share PGA lead at difficult Aronimink
-
Drake drops three albums at once

-
 Boeing confirms China commitment to buy 200 aircraft
Boeing confirms China commitment to buy 200 aircraft
-
Knicks forward Anunoby trains as NBA Eastern Conference finals loom

-
 American McNealy grabs PGA lead at difficult Aronimink
American McNealy grabs PGA lead at difficult Aronimink
-
Substitute 'keeper sends Saint-Etienne into promotion play-off

-
 Sinner's bid to reach Italian Open final held up by Roman rain
Sinner's bid to reach Italian Open final held up by Roman rain
-
Aston Villa humble Liverpool to secure Champions League qualification

-
 US says Iran-backed militia commander planned Jewish site attacks
US says Iran-backed militia commander planned Jewish site attacks
-
Bolivia unrest continues despite government deal with miners

-
 Scheffler slams 'absurd' PGA pin locations
Scheffler slams 'absurd' PGA pin locations
-
New deadly Ebola outbreak hits DR Congo, 1 dead in Uganda

-
 Democrats accuse Trump of stock trade corruption
Democrats accuse Trump of stock trade corruption
-
'Beyond the Oscar': Travolta gets surprise Cannes prize

-
 Israel, Lebanon say extending ceasefire despite new strikes
Israel, Lebanon say extending ceasefire despite new strikes
-
Potgieter grabs early PGA lead at difficult Aronimink

-
 Prosecutors seek death penalty for US man charged with killing Israeli embassy staffers
Prosecutors seek death penalty for US man charged with killing Israeli embassy staffers
-
Judge declares mistrial in Weinstein sex assault case

-
 Canada takes key step towards new oil pipeline
Canada takes key step towards new oil pipeline
-
Iranian filmmaker Farhadi condemns Middle East war, protest massacres

-
 'Better than the Oscar': John Travolta gets surprise Cannes prize
'Better than the Oscar': John Travolta gets surprise Cannes prize
-
Marsh muscle motors Lucknow to victory over Chennai

-
 Judge declares mistrial in Weinstein case as jury fails to reach verdict
Judge declares mistrial in Weinstein case as jury fails to reach verdict
-
Eurovision finalists tune up as boycotting Spain digs in
-
 Indonesia's first giant panda is set to charm the public
Indonesia's first giant panda is set to charm the public
-
Cheer and tears as African refugee rap film 'Congo Boy' charms Cannes

-
 Norwegian Ruud rolls into Italian Open final, Sinner set for Medvedev clash
Norwegian Ruud rolls into Italian Open final, Sinner set for Medvedev clash
-
Bolivia government says deal reached with protesting miners

-
 Showdowns and spycraft on Trump-Xi summit sidelines
Showdowns and spycraft on Trump-Xi summit sidelines
-
Smalley seizes PGA lead with Matsuyama making a charge


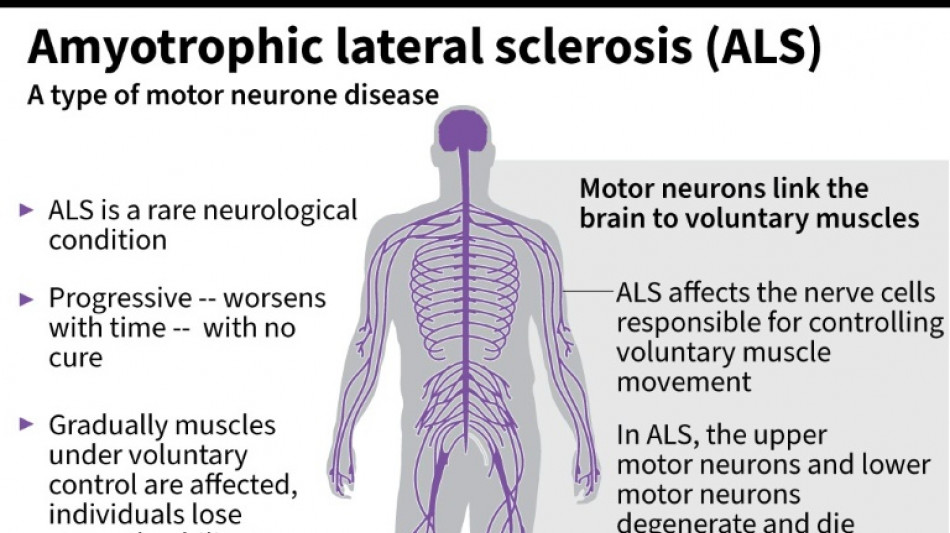
Every month counts: European ALS patients want new drugs
Olivier Goy is running out of time.
The French entrepreneur was diagnosed in 2020 with amyotrophic lateral sclerosis (ALS) -- the incurable neurodegenerative disease that normally claims the lives of patients within three to five years.
There are new treatments that have given patients hope of being able to extend their lives by an invaluable few months, but the approval process in Europe is taking time, infuriating desperate patients.
"When you are certain to die soon, patients and some doctors are ready to take some risks," Goy told AFP.
In response to the lack of new treatments in his native France, the founder of the fintech start-up October spends 3,000 euros ($3,180) every month to buy the ingredients to make his own drugs.
ALS, also known as Lou Gehrig's disease, attacks the motor nerve cells in the brain and spinal cord, progressively paralysing muscles until patients cannot walk, eat, speak or breathe.
Around one in 10,000 people have the disease in the EU, according to the European Medicines Agency.
The drug Riluzole, which has been available in Europe and the UK since the 1990s, is capable of prolonging the lives of patients by around three months.
But otherwise, no new treatment has been approved in Europe for more than two decades.
- 'First hope in 20 years' -
A new treatment called AMX0035 was given the green light in the United States and Canada last year.
"It is the first hope we have had in 20 years: the first drug which is aimed at everyone and which had results" suggesting up to six months in added life expectancy, said Sabine Turgeman, head of the French Association for Research into ALS.
But the extent of the benefits of AMX0035 remains unclear. The US Food and Drug Administration approved the drug, sold under the name Relyvrio, based on the results of a single Phase 2 trial that involved just 137 participants.
The drug's developer, Amylyx Pharmaceuticals, is conducting larger, more comprehensive trials, with results expected in 2024.
Amylyx said earlier this month that the European Union's drug watchdog EMA is reviewing its submission for approval and it expects a decision in the first half of this year.
But for those with the disease, every delay represents a significant amount of the time they have left.
"It's not going fast enough," Turgeman said. "This disease is not on bureaucratic time".
For European patients who cannot afford to import their own ingredients like Goy, the only way to get access to new treatments is to join a clinical trial.
But such trials have very specific criteria for selection -- and even if a patient gets in, there is a chance they will be in the group given a placebo.
- 'Totally abandoned' -
Given how swiftly the disease progresses, patients and families are pressing for more options.
"We feel totally abandoned," said Sophie Garofalo, whose brother was diagnosed with ALS five years ago.
His family tried to enter him into clinical trials, "but either he does not meet the criteria, or the trials have already started," she said.
"He is ready to take anything, try everything".
French pharmaceutical company AB Science is developing another potential treatment using the drug masitinib, which initial results suggest could add months to the lives of patients.
The firm's CEO Alain Moussy said that because "time is very limited" for ALS patients, there should be more flexibility in the approval system.
"What degree of risk should be taken? That's for the health agencies to answer -- but they can guided by policymakers and patients," he said.
H.Seidel--BTB



