
-
 French president Macron heads to South Korea after Japan visit
French president Macron heads to South Korea after Japan visit
-
Armenia's underground salt clinic at centre of alternative medicine debate

-
 'Muted' international response as Senegal enacts same-sex relations law
'Muted' international response as Senegal enacts same-sex relations law
-
Slow boat to Ilulissat: long nights on Greenland's last ferry

-
 Wemby rampant again as Spurs rack up 10th straight win
Wemby rampant again as Spurs rack up 10th straight win
-
Ukrainian death metal band growls against Russia's war

-
 Iran fires missiles at Israel after Trump threatens weeks of strikes
Iran fires missiles at Israel after Trump threatens weeks of strikes
-
Surging 'Jewish terrorism' in West Bank condemned but unpunished

-
 England's Brook, Bethell warned after New Zealand nightclub incident
England's Brook, Bethell warned after New Zealand nightclub incident
-
What's real anymore? AI warps truth of Middle East war

-
 Europe to negotiate with NASA on lunar missions: ESA
Europe to negotiate with NASA on lunar missions: ESA
-
Trump tells US that Iran war victory near, but vows big strikes

-
 Poppies offer hope in fire-scarred Los Angeles
Poppies offer hope in fire-scarred Los Angeles
-
Trump says Iran war almost over, warns of weeks more heavy strikes

-
 Oil rallies, stocks tumble as Trump says US to hammer Iran further
Oil rallies, stocks tumble as Trump says US to hammer Iran further
-
US Republicans announce deal to end partial government shutdown

-
 Trump tells Americans that Iran war ending as popularity dips
Trump tells Americans that Iran war ending as popularity dips
-
7.4-magnitude quake off Indonesia kills one, tsunami warning lifted

-
 Bordeaux-Begles' Van Rensburg 'not thinking' about Champions Cup double
Bordeaux-Begles' Van Rensburg 'not thinking' about Champions Cup double
-
US automakers report mixed sales as car market awaits war impact

-
 Astronauts begin NASA lunar mission after climactic blast-off
Astronauts begin NASA lunar mission after climactic blast-off
-
Astronauts blast off for historic US lunar journey

-
 Embattled Woods won't captain 2027 Ryder Cup team: PGA of America
Embattled Woods won't captain 2027 Ryder Cup team: PGA of America
-
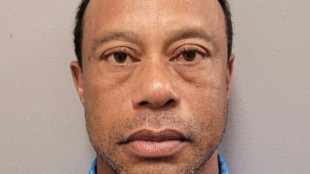
Judge allows Woods to travel overseas for treatment
-
 Chelsea's Bompastor furious as Arsenal reach women's Champions League semis
Chelsea's Bompastor furious as Arsenal reach women's Champions League semis
-
US lifts sanctions on Venezuelan interim leader Delcy Rodriguez

-
 Arsenal resist Chelsea rally to reach women's Champions League semis
Arsenal resist Chelsea rally to reach women's Champions League semis
-
Defending champ Pegula wins WTA Charleston opener

-
 New frog species carrying eggs on back discovered in Peru
New frog species carrying eggs on back discovered in Peru
-
Benfica winger Prestianni denies 'ugly' racism claims

-
 Tuchel casts doubt on Foden's World Cup chances
Tuchel casts doubt on Foden's World Cup chances
-
Slot hoping Salah can still burnish Liverpool legacy

-
 Astronauts strapped in for historic US lunar launch
Astronauts strapped in for historic US lunar launch
-
Top World Bank official 'extremely concerned' by fallout of Iran war

-
 'Wake-up call': Megan Thee Stallion falls ill during Broadway show
'Wake-up call': Megan Thee Stallion falls ill during Broadway show
-
Canada's defense enters new phase, Arctic in focus: top military officer

-
 France charges man over failed attack on US bank
France charges man over failed attack on US bank
-
Bayern reach women's Champions League semis after late show sinks United

-
 SpaceX files to go public, paving way for record stock offering
SpaceX files to go public, paving way for record stock offering
-
Delhi make winning start to IPL as Rizvi downs LSG

-
 Final ticket sales phase begins for FIFA World Cup
Final ticket sales phase begins for FIFA World Cup
-
Supreme Court skeptical of Trump bid to end birthright citizenship

-
 Tractors roll through Vienna as farmers protest
Tractors roll through Vienna as farmers protest
-
PGA Tour, Masters chairman support Tiger recovery pause

-
 World Cup winner Goetze extends contract at Frankfurt
World Cup winner Goetze extends contract at Frankfurt
-
SpaceX files securities documents to go public: source

-
 Armenia cannot be in both EU and Russian customs bloc, Putin says
Armenia cannot be in both EU and Russian customs bloc, Putin says
-
Supreme Court hears landmark citizenship case -- with Trump in audience

-
 Chelsea announce record pre-tax loss of £262.4 million
Chelsea announce record pre-tax loss of £262.4 million
-
Stocks rally, oil drops on Mideast war optimism


Biogen pulls controversial Alzheimer's drug Aduhelm
A controversial Alzheimer's drug that was trumpeted as the first to ever treat the cognitive decline associated with the devastating brain disorder has been pulled from the market, its maker Biogen announced Wednesday.
The US Food and Drug Administration awarded accelerated approval to Aduhelm in June 2021, a decision that was highly contentious at the time because the agency overruled its own independent advisors, who found there was insufficient evidence of benefit.
At least three of the 11-member independent committee that voted unanimously against recommending the drug subsequently resigned, and US congressional investigators slammed the accelerated approval as "rife with irregularities."
Biogen said it was discontinuing Aduhelm to put more resources into Leqembi, a newer Alzheimer's medicine that was fully approved last year under the traditional regulatory pathway.
"When searching for new medicines, one breakthrough can be the foundation that triggers future medicines to be developed," said Christopher Viehbacher, president and CEO of the Cambridge, Massachusetts-based biotech firm.
"Aduhelm was that groundbreaking discovery that paved the way for a new class of drugs and reinvigorated investments in the field."
Aduhelm, a monoclonal antibody that targets the build-up of a protein called amyloid beta in the brain tissue which is thought to be a cause of Alzheimer's, was tested in two late-stage human trials.
It showed a reduction in cognitive decline in one of the studies, but not the other.
According to a congressional report from December 2022, the FDA "considered Aduhelm under the traditional approval pathway used for most drugs for nine months, before abruptly changing course and granting approval under the accelerated approval pathway after a three-week review period."
The report said that FDA interactions with Biogen were "atypical" and included a failure to properly document contacts between agency staff and the drug maker.
The FDA and Biogen had also "inappropriately collaborated" on a joint briefing document for a key advisory committee, it said.
"FDA's approval process was rife with irregularities."
As for Biogen, the report said the company "viewed Aduhelm as an unprecedented financial opportunity -- estimating a potential peak revenue of $18 billion per year."
The congressional panel pointed to an "unjustifiably high price" for Aduhelm of $56,000 a year for patients.
Biogen's Leqembi, which it co-manufactures with Eisai of Japan, is now the only US approved treatment for Alzheimer's. It also targets amyloid beta and has been found to modestly reduce cognitive decline in patients with early stage disease.
Donanemab, developed by Eli Lilly, could be next to get the green light after performing similarly in clinical trials.
Alzheimer's is the most common form of dementia. More than one in nine people over 65 develop the condition, which worsens over time, robbing them of their memories and independence, according to the US Alzheimer's Association.
W.Lapointe--BTB



