
-
 Marsh muscle motors Lucknow to victory over Chennai
Marsh muscle motors Lucknow to victory over Chennai
-
Judge declares mistrial in Weinstein case as jury fails to reach verdict

-
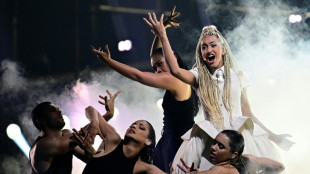 Eurovision finalists tune up as boycotting Spain digs in
Eurovision finalists tune up as boycotting Spain digs in
-
Indonesia's first giant panda is set to charm the public

-
 Cheer and tears as African refugee rap film 'Congo Boy' charms Cannes
Cheer and tears as African refugee rap film 'Congo Boy' charms Cannes
-
Norwegian Ruud rolls into Italian Open final, Sinner set for Medvedev clash

-
 Bolivia government says deal reached with protesting miners
Bolivia government says deal reached with protesting miners
-
Showdowns and spycraft on Trump-Xi summit sidelines

-
 Smalley seizes PGA lead with Matsuyama making a charge
Smalley seizes PGA lead with Matsuyama making a charge
-
Acosta quickest in practice for Catalan MotoGP

-
 Nuno wants VAR 'consistency' as West Ham fight to avoid relegation
Nuno wants VAR 'consistency' as West Ham fight to avoid relegation
-
Vingegaard powers to maiden Giro stage victory

-
 Iran to hold pre-World Cup training camp in Turkey: media
Iran to hold pre-World Cup training camp in Turkey: media
-
US scraps deployment of 4,000 troops to Poland

-
 Ukraine vows more strikes on Russia after attack on Kyiv kills 24
Ukraine vows more strikes on Russia after attack on Kyiv kills 24
-
Bayern veteran Neuer signs one-year contract extension

-
 Ukraine can down Russian drones en masse. But missiles are a problem
Ukraine can down Russian drones en masse. But missiles are a problem
-
Israeli strikes wound dozens in Lebanon as talks in US enter second day

-
 'Everybody wants Hearts to win', says Celtic's O'Neill ahead of title decider
'Everybody wants Hearts to win', says Celtic's O'Neill ahead of title decider
-
Scheffler stumbles from share of lead at windy PGA

-
 New deadly Ebola outbreak hits DR Congo
New deadly Ebola outbreak hits DR Congo
-
Farke calls for Leeds owners to match his ambition

-
 Zverev pulls out of home event in Hamburg with back injury
Zverev pulls out of home event in Hamburg with back injury
-
Xi, Trump eke small wins from talks but no major deals: analysts

-
 De Ligt to miss World Cup after back surgery
De Ligt to miss World Cup after back surgery
-
England's Rice braces for 'hate and love' at World Cup

-
 Milan Fashion Week says will ask brands not to show fur
Milan Fashion Week says will ask brands not to show fur
-
French-German tank maker KNDS to push ahead with IPO

-
 Man City campaign a success regardless of trophies: Guardiola
Man City campaign a success regardless of trophies: Guardiola
-
'World's oldest dog' contender dies in France aged 30

-
 No.1 Scheffler opens with bogey to fall from share of PGA lead
No.1 Scheffler opens with bogey to fall from share of PGA lead
-
Carrick says Man Utd future to be decided 'pretty soon'

-
 'Out of shape' Lukaku named in Belgium World Cup squad
'Out of shape' Lukaku named in Belgium World Cup squad
-
Hearts ready to 'rip up the script' in Celtic title showdown

-
 X pledges crackdown on illegal content in UK
X pledges crackdown on illegal content in UK
-
Possible contenders in UK Labour Party leadership race

-
 Germany's Merz says wouldn't advise young people to move to US
Germany's Merz says wouldn't advise young people to move to US
-
Israel strikes Lebanon as talks in US enter second day

-
 Kyiv in mourning after 24 killed as Ukraine, Russia swap POWs
Kyiv in mourning after 24 killed as Ukraine, Russia swap POWs
-
Beckham becomes first British billionaire sportsman

-
 Aussie star, Danish clubbing ode through to Eurovision final
Aussie star, Danish clubbing ode through to Eurovision final
-
German Oscar winner Huller feels war guilt 'every day'

-
 Thai lawmakers vote to revive clean air bill
Thai lawmakers vote to revive clean air bill
-
Bayern warn that Canada's Davies struggling to be fit for World Cup

-
 Long-serving Coleman to end Everton career at end of season
Long-serving Coleman to end Everton career at end of season
-
Energy-hungry German industries in decline since Ukraine war: data

-
 Gordon may have made last Newcastle appearance: Howe
Gordon may have made last Newcastle appearance: Howe
-
Denmark's Queen Margrethe has angioplasty in hospital: palace

-
 Civilians caught in war of drones in eastern DR Congo
Civilians caught in war of drones in eastern DR Congo
-
French city reels from teen killing in drug-linked shooting


Pfizer to seek US authorization for third Covid shot in children
Pfizer and BioNTech on Thursday announced positive results from a clinical trial on the safety and immune response of a third dose of their Covid vaccine in children aged five through 11, adding they would soon seek regulatory authorization.
Third doses of the vaccine are recommended for those aged 12 and up, and a fourth dose was recently recommended for people over 50.
Younger children -- except for those with immune compromising conditions -- have not been eligible for the third, making them more susceptible to infection from Omicron and its BA.2 subvariant.
BA.2 is now the globally dominant strain, and is behind a current spike in cases in the northeastern United States.
In the phase 2/3 trial, the companies analyzed data from 140 children aged five through 11, approximately six months after the second dose.
The dosage in this group is 10 micrograms, which was selected for safety reasons as children are more susceptible to side effects. The dose for those 12 and up is 30 micrograms.
Across the 140 children analyzed, the third dose was well tolerated, revealing no new safety concerns.
They also analyzed blood sera from a group of 30 individuals, finding that a third dose caused a 36-fold increase in levels of infection-blocking neutralizing antibodies against Omicron, compared to two doses.
Pfizer and BioNTech plan to soon submit the data to the US Food and Drug Administration (FDA), the European Medicines Agency and other regulatory agencies.
Most countries, including the United States, haven't yet authorized Covid vaccines for infants and very young children.
Last month, Moderna said it was pursuing approval for its vaccine in children aged six months through five years, using a two-dose regimen.
Pfizer's vaccine for this group was meant to be considered by the FDA in February but the agency postponed the meeting, because it wanted more data on how it would perform with three doses.
D.Schneider--BTB



