-
 McNealy and Smalley share PGA lead at difficult Aronimink
McNealy and Smalley share PGA lead at difficult Aronimink
-
Drake drops three albums at once

-
 Boeing confirms China commitment to buy 200 aircraft
Boeing confirms China commitment to buy 200 aircraft
-
Knicks forward Anunoby trains as NBA Eastern Conference finals loom

-
 American McNealy grabs PGA lead at difficult Aronimink
American McNealy grabs PGA lead at difficult Aronimink
-
Substitute 'keeper sends Saint-Etienne into promotion play-off

-
 Sinner's bid to reach Italian Open final held up by Roman rain
Sinner's bid to reach Italian Open final held up by Roman rain
-
Aston Villa humble Liverpool to secure Champions League qualification

-
 US says Iran-backed militia commander planned Jewish site attacks
US says Iran-backed militia commander planned Jewish site attacks
-
Bolivia unrest continues despite government deal with miners

-
 Scheffler slams 'absurd' PGA pin locations
Scheffler slams 'absurd' PGA pin locations
-
New deadly Ebola outbreak hits DR Congo, 1 dead in Uganda

-
 Democrats accuse Trump of stock trade corruption
Democrats accuse Trump of stock trade corruption
-
'Beyond the Oscar': Travolta gets surprise Cannes prize

-
 Israel, Lebanon say extending ceasefire despite new strikes
Israel, Lebanon say extending ceasefire despite new strikes
-
Potgieter grabs early PGA lead at difficult Aronimink

-
 Prosecutors seek death penalty for US man charged with killing Israeli embassy staffers
Prosecutors seek death penalty for US man charged with killing Israeli embassy staffers
-
Judge declares mistrial in Weinstein sex assault case

-
 Canada takes key step towards new oil pipeline
Canada takes key step towards new oil pipeline
-
Iranian filmmaker Farhadi condemns Middle East war, protest massacres

-
 'Better than the Oscar': John Travolta gets surprise Cannes prize
'Better than the Oscar': John Travolta gets surprise Cannes prize
-
Marsh muscle motors Lucknow to victory over Chennai

-
 Judge declares mistrial in Weinstein case as jury fails to reach verdict
Judge declares mistrial in Weinstein case as jury fails to reach verdict
-
Eurovision finalists tune up as boycotting Spain digs in
-
 Indonesia's first giant panda is set to charm the public
Indonesia's first giant panda is set to charm the public
-
Cheer and tears as African refugee rap film 'Congo Boy' charms Cannes

-
 Norwegian Ruud rolls into Italian Open final, Sinner set for Medvedev clash
Norwegian Ruud rolls into Italian Open final, Sinner set for Medvedev clash
-
Bolivia government says deal reached with protesting miners

-
 Showdowns and spycraft on Trump-Xi summit sidelines
Showdowns and spycraft on Trump-Xi summit sidelines
-
Smalley seizes PGA lead with Matsuyama making a charge

-
 Acosta quickest in practice for Catalan MotoGP
Acosta quickest in practice for Catalan MotoGP
-
Nuno wants VAR 'consistency' as West Ham fight to avoid relegation

-
 Vingegaard powers to maiden Giro stage victory
Vingegaard powers to maiden Giro stage victory
-
Iran to hold pre-World Cup training camp in Turkey: media

-
 US scraps deployment of 4,000 troops to Poland
US scraps deployment of 4,000 troops to Poland
-
Ukraine vows more strikes on Russia after attack on Kyiv kills 24

-
 Bayern veteran Neuer signs one-year contract extension
Bayern veteran Neuer signs one-year contract extension
-
Ukraine can down Russian drones en masse. But missiles are a problem

-
 Israeli strikes wound dozens in Lebanon as talks in US enter second day
Israeli strikes wound dozens in Lebanon as talks in US enter second day
-
'Everybody wants Hearts to win', says Celtic's O'Neill ahead of title decider

-
 Scheffler stumbles from share of lead at windy PGA
Scheffler stumbles from share of lead at windy PGA
-
New deadly Ebola outbreak hits DR Congo

-
 Farke calls for Leeds owners to match his ambition
Farke calls for Leeds owners to match his ambition
-
Zverev pulls out of home event in Hamburg with back injury

-
 Xi, Trump eke small wins from talks but no major deals: analysts
Xi, Trump eke small wins from talks but no major deals: analysts
-
De Ligt to miss World Cup after back surgery

-
 England's Rice braces for 'hate and love' at World Cup
England's Rice braces for 'hate and love' at World Cup
-
Milan Fashion Week says will ask brands not to show fur

-
 French-German tank maker KNDS to push ahead with IPO
French-German tank maker KNDS to push ahead with IPO
-
Man City campaign a success regardless of trophies: Guardiola


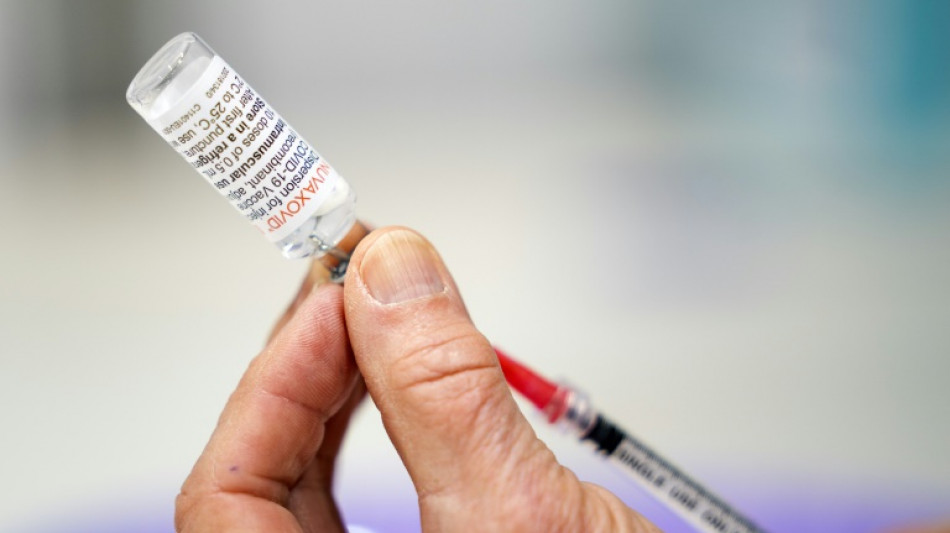
FDA experts weigh authorizing Novavax Covid-19 vaccine in US
A panel of experts convened by the US drug regulator was meeting Tuesday to consider authorizing the Novavax Covid-19 shot, a late runner in the fight against the virus that could nonetheless play a role in overcoming vaccine hesitancy.
Three vaccines are currently approved in the United States: Pfizer and Moderna, which are based on messenger RNA, and Johnson and Johnson, an adenovirus vector vaccine.
But the last of these, the J&J vaccine, was recently restricted in the US after being linked to a rare but serious clotting condition, especially in women of reproductive age.
It is now only recommended for adults who cannot access Pfizer or Moderna for medical or other serious reasons.
The Novavax vaccine was an early frontrunner in the vaccine race, but fell behind after being hit by manufacturing and regulatory delays.
Though the company is American, the US is one of the few major markets where it hasn't yet received authorization -- the EU, UK, Canada, Australia are among many that have already given it the green light.
Officials hope that the shot, which is based on synthetic proteins, could provide an alternative to the mRNA vaccines for people still hesitant.
"We do have a problem with vaccine uptake that is very serious in the United States," Peter Marks, a senior scientist for the Food and Drug administration, said at the start of the meeting.
"And anything we can do to get people more comfortable to be able to accept these potentially life saving medical products, is something that we feel we are compelled to do."
Of the various vaccine technologies, mRNA has been subject to the most misinformation efforts.
Novavax's vaccine was found to be 90 percent effective against symptomatic cases of the disease, in trials conducted before the appearance of the Omicron variant, according to the FDA.
But six cases of myocarditis, an inflammation of the heart muscle, were detected in the group that received the vaccine, against one case in the placebo group, in a trial of around 40,000 people.
Novavax says there is insufficient evidence to establish a causal relationship between the cases of myocarditis and the vaccine.
The FDA voiced concern over the myocarditis link on Friday, sending Novavax shares to drop 20 percent on the New York Stock Exchange. And trading of Novavax stock was halted on Monday pending news from the FDA panel.
Known as a protein subunit vaccine, Novavax is administered in two doses.
It uses a synthetic version of the virus' spike protein to evoke an immune response.
The same technique is used in vaccines against whooping cough, meningococcal meningitis and hepatitis B.
M.Furrer--BTB


