-
 McIlroy, back in PGA hunt, blames bad setup for lead logjam
McIlroy, back in PGA hunt, blames bad setup for lead logjam
-
Kubo vows to lead Japan at World Cup with Mitoma out

-
 McNealy and Smalley share PGA lead at difficult Aronimink
McNealy and Smalley share PGA lead at difficult Aronimink
-
Drake drops three albums at once

-
 Boeing confirms China commitment to buy 200 aircraft
Boeing confirms China commitment to buy 200 aircraft
-
Knicks forward Anunoby trains as NBA Eastern Conference finals loom

-
 American McNealy grabs PGA lead at difficult Aronimink
American McNealy grabs PGA lead at difficult Aronimink
-
Substitute 'keeper sends Saint-Etienne into promotion play-off

-
 Sinner's bid to reach Italian Open final held up by Roman rain
Sinner's bid to reach Italian Open final held up by Roman rain
-
Aston Villa humble Liverpool to secure Champions League qualification

-
 US says Iran-backed militia commander planned Jewish site attacks
US says Iran-backed militia commander planned Jewish site attacks
-
Bolivia unrest continues despite government deal with miners

-
 Scheffler slams 'absurd' PGA pin locations
Scheffler slams 'absurd' PGA pin locations
-
New deadly Ebola outbreak hits DR Congo, 1 dead in Uganda

-
 Democrats accuse Trump of stock trade corruption
Democrats accuse Trump of stock trade corruption
-
'Beyond the Oscar': Travolta gets surprise Cannes prize

-
 Israel, Lebanon say extending ceasefire despite new strikes
Israel, Lebanon say extending ceasefire despite new strikes
-
Potgieter grabs early PGA lead at difficult Aronimink

-
 Prosecutors seek death penalty for US man charged with killing Israeli embassy staffers
Prosecutors seek death penalty for US man charged with killing Israeli embassy staffers
-
Judge declares mistrial in Weinstein sex assault case

-
 Canada takes key step towards new oil pipeline
Canada takes key step towards new oil pipeline
-
Iranian filmmaker Farhadi condemns Middle East war, protest massacres

-
 'Better than the Oscar': John Travolta gets surprise Cannes prize
'Better than the Oscar': John Travolta gets surprise Cannes prize
-
Marsh muscle motors Lucknow to victory over Chennai

-
 Judge declares mistrial in Weinstein case as jury fails to reach verdict
Judge declares mistrial in Weinstein case as jury fails to reach verdict
-
Eurovision finalists tune up as boycotting Spain digs in
-
 Indonesia's first giant panda is set to charm the public
Indonesia's first giant panda is set to charm the public
-
Cheer and tears as African refugee rap film 'Congo Boy' charms Cannes

-
 Norwegian Ruud rolls into Italian Open final, Sinner set for Medvedev clash
Norwegian Ruud rolls into Italian Open final, Sinner set for Medvedev clash
-
Bolivia government says deal reached with protesting miners

-
 Showdowns and spycraft on Trump-Xi summit sidelines
Showdowns and spycraft on Trump-Xi summit sidelines
-
Smalley seizes PGA lead with Matsuyama making a charge

-
 Acosta quickest in practice for Catalan MotoGP
Acosta quickest in practice for Catalan MotoGP
-
Nuno wants VAR 'consistency' as West Ham fight to avoid relegation

-
 Vingegaard powers to maiden Giro stage victory
Vingegaard powers to maiden Giro stage victory
-
Iran to hold pre-World Cup training camp in Turkey: media

-
 US scraps deployment of 4,000 troops to Poland
US scraps deployment of 4,000 troops to Poland
-
Ukraine vows more strikes on Russia after attack on Kyiv kills 24

-
 Bayern veteran Neuer signs one-year contract extension
Bayern veteran Neuer signs one-year contract extension
-
Ukraine can down Russian drones en masse. But missiles are a problem

-
 Israeli strikes wound dozens in Lebanon as talks in US enter second day
Israeli strikes wound dozens in Lebanon as talks in US enter second day
-
'Everybody wants Hearts to win', says Celtic's O'Neill ahead of title decider

-
 Scheffler stumbles from share of lead at windy PGA
Scheffler stumbles from share of lead at windy PGA
-
New deadly Ebola outbreak hits DR Congo

-
 Farke calls for Leeds owners to match his ambition
Farke calls for Leeds owners to match his ambition
-
Zverev pulls out of home event in Hamburg with back injury

-
 Xi, Trump eke small wins from talks but no major deals: analysts
Xi, Trump eke small wins from talks but no major deals: analysts
-
De Ligt to miss World Cup after back surgery

-
 England's Rice braces for 'hate and love' at World Cup
England's Rice braces for 'hate and love' at World Cup
-
Milan Fashion Week says will ask brands not to show fur


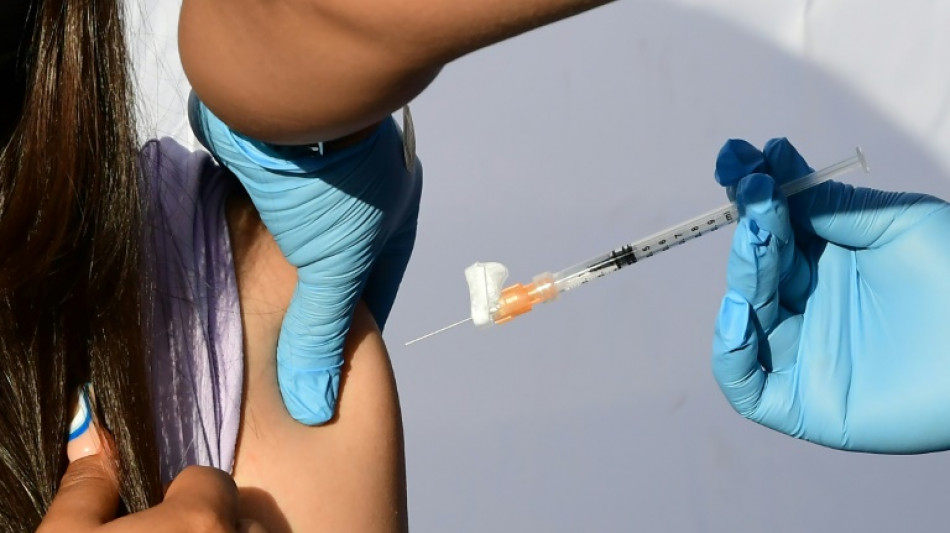
US panel weighs authorizing Covid vaccines for youngest children
After months of anxious wait for some parents, a panel of experts convened by the US Food and Drug Administration will meet Wednesday to weigh recommending Covid vaccines for the nation's youngest children.
Children under five are the only age group not yet eligible for Covid immunization in the United States and most other countries. If, as expected, panelists vote in favor of greenlighting the Pfizer and Moderna vaccines, formal authorizations should follow quickly, with the first shots in arms expected by next week.
Ahead of the meeting, the FDA posted its independent analyses of the two drugs, deeming both safe and effective. It also said there was a pressing unmet need to inoculate children in the age group since their rates of hospitalization and death "are higher than among children and adolescents 5-17 years of age."
Both vaccines are based on messenger RNA technology, which delivers genetic code for the coronavirus spike protein to human cells, training the immune system to be ready for when it encounters the real virus.
Pfizer is seeking authorization for three doses at three micrograms given to ages six months through four years, while Moderna has asked for the FDA to approve its vaccine as two doses of a higher 30 micrograms, for ages six months through five years.
They were tested in trials of thousands of children where they were found to have similar levels of mild effects to older age groups and triggered similar levels of antibodies.
Efficacy against infection was higher for Pfizer, with the company placing it at 80 percent compared to Moderna's estimates of 51 percent in the six-month-old to age two group and 37 percent in the two to five years age group.
But the figures are provisional and Moderna is studying adding a third dose later that may increase its figures.
There are some 20 million US children aged four years and under. If the FDA-appointed experts recommend the two vaccines, then the matter will go to another committee convened by the Centers for Disease Control and Prevention for a final say.
White House officials last week said the rollout of millions of shots at pharmacies and doctors' offices could begin as soon as June 21, following the Juneteenth holiday on June 20.
Of the total US Covid deaths, 480 have come in children under five, according to the latest official data.
Obesity, neurological disorders and asthma are associated with increased risk of severe disease, "however, a majority of children hospitalized for Covid-19 have no underlying medical conditions," the FDA said in a document.
Children can also go on to contract multisystem inflammatory syndrome in children (MIS-C), a rare but serious post-viral condition.
Data on long Covid in children is sparse, but the FDA's document cited a national survey in the United Kingdom which found that "among children ages two to 11 years who tested positive for COVID-19, 7.2 percent reported continued symptoms at 12 weeks."
E.Schubert--BTB


