-
 Trump urges Bruce Springsteen boycott in social media rant
Trump urges Bruce Springsteen boycott in social media rant
-
US banks in Paris tighten security, order remote work over pro-Iran threat

-
 Israeli politicians, ex-security officials slam 'Jewish terrorism' in West Bank
Israeli politicians, ex-security officials slam 'Jewish terrorism' in West Bank
-
Bashir retains England 'ambition' despite Ashes snub

-
 US trade deficit widens less than forecast as tariff turmoil persists
US trade deficit widens less than forecast as tariff turmoil persists
-
UEFA chief Ceferin warns Italy could lose Euro 2032 without stadium improvements

-
 Italy's football chief resigns after World Cup disaster
Italy's football chief resigns after World Cup disaster
-
Edoardo Molinari named European vice-captain for Ryder Cup

-
 'Extraordinary news': Dutch recover stolen gold Romanian helmet
'Extraordinary news': Dutch recover stolen gold Romanian helmet
-
France considers reform for New Caledonia

-
 UK foreign minister stresses 'urgent need' to reopen Hormuz strait
UK foreign minister stresses 'urgent need' to reopen Hormuz strait
-
Macron says Trump marriage jibe does not 'merit response'

-
 Russia will send second ship with oil to Cuba: minister
Russia will send second ship with oil to Cuba: minister
-
Belgian bishop takes on Vatican with push to ordain married men

-
 Oil rallies, stocks drop as Trump dampens Mideast hopes
Oil rallies, stocks drop as Trump dampens Mideast hopes
-
Nexperia's China unit nears fully local production of chips: company sources

-
 Indonesia issues fresh summons for Google, Meta over teen social media ban
Indonesia issues fresh summons for Google, Meta over teen social media ban
-
Japan axe coach Nielsen 12 days after winning Women's Asian Cup

-
 French President Macron lands in South Korea after Japan visit
French President Macron lands in South Korea after Japan visit
-
India's says defence exports hit 'all-time high' of $4 bn

-
 Nielsen leaves as Japan coach weeks after winning Women's Asian Cup
Nielsen leaves as Japan coach weeks after winning Women's Asian Cup
-
Too bright: Seoul to dim digital billboards after complaints

-
 Iran vows 'crushing' attacks on US after Trump threats
Iran vows 'crushing' attacks on US after Trump threats
-
Women's Asian Cup finalists accuse governing body over equal money

-
 French president Macron heads to South Korea after Japan visit
French president Macron heads to South Korea after Japan visit
-
Armenia's underground salt clinic at centre of alternative medicine debate

-
 'Muted' international response as Senegal enacts same-sex relations law
'Muted' international response as Senegal enacts same-sex relations law
-
Slow boat to Ilulissat: long nights on Greenland's last ferry

-
 Wemby rampant again as Spurs rack up 10th straight win
Wemby rampant again as Spurs rack up 10th straight win
-
Ukrainian death metal band growls against Russia's war

-
 Iran fires missiles at Israel after Trump threatens weeks of strikes
Iran fires missiles at Israel after Trump threatens weeks of strikes
-
Surging 'Jewish terrorism' in West Bank condemned but unpunished

-
 England's Brook, Bethell warned after New Zealand nightclub incident
England's Brook, Bethell warned after New Zealand nightclub incident
-
What's real anymore? AI warps truth of Middle East war

-
 Europe to negotiate with NASA on lunar missions: ESA
Europe to negotiate with NASA on lunar missions: ESA
-
Trump tells US that Iran war victory near, but vows big strikes

-
 Poppies offer hope in fire-scarred Los Angeles
Poppies offer hope in fire-scarred Los Angeles
-
Trump says Iran war almost over, warns of weeks more heavy strikes

-
 Oil rallies, stocks tumble as Trump says US to hammer Iran further
Oil rallies, stocks tumble as Trump says US to hammer Iran further
-
US Republicans announce deal to end partial government shutdown

-
 Trump tells Americans that Iran war ending as popularity dips
Trump tells Americans that Iran war ending as popularity dips
-
7.4-magnitude quake off Indonesia kills one, tsunami warning lifted

-
 Bordeaux-Begles' Van Rensburg 'not thinking' about Champions Cup double
Bordeaux-Begles' Van Rensburg 'not thinking' about Champions Cup double
-
Valsoft Corporation Strengthens Its Retail Software Portfolio with the Acquisition of NedFox

-
 Side-by-Side Conference Hosts Close to 800 Participants in Malta to Advance Women's Health Care
Side-by-Side Conference Hosts Close to 800 Participants in Malta to Advance Women's Health Care
-
U.S. Polo Assn. Supports Division I National Intercollegiate Championship, Showcasing the Future of the Sport of Polo

-
 US automakers report mixed sales as car market awaits war impact
US automakers report mixed sales as car market awaits war impact
-
Astronauts begin NASA lunar mission after climactic blast-off

-
 Astronauts blast off for historic US lunar journey
Astronauts blast off for historic US lunar journey
-
Embattled Woods won't captain 2027 Ryder Cup team: PGA of America


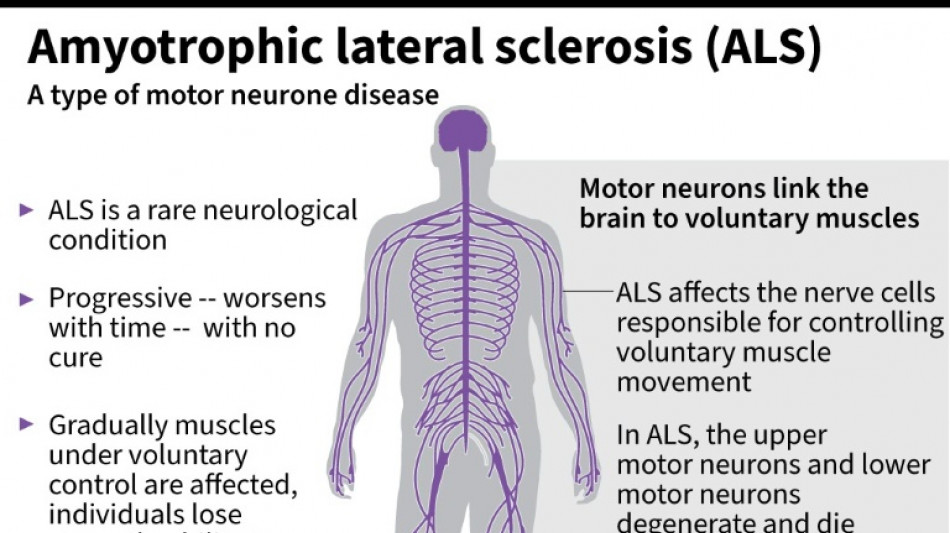
US company withdraws ALS drug after it fails in trial
Amylyx Pharmaceuticals announced Thursday it was withdrawing its approved treatment against the deadly neurodegenerative disease ALS after clinical data found no evidence the drug worked.
In a statement, the US company said it would discontinue its market authorizations for Relyvrio/Albrioza, using the brand names of the medicine in the US and Canadian markets.
"While this is a difficult moment for the ALS community, we reached this path forward in partnership with the stakeholders who will be impacted and in line with our steadfast commitment to people living with ALS and other neurodegenerative diseases," said the company's co-CEOs Joshua Cohen and Justin Klee in a statement.
The company also said it was reducing its workforce "by approximately 70 percent" as it focused on another experimental drug for use against ALS, and on repurposing Relyvrio for other conditions. It added it would continue to make Relyvrio available for patients who wish to keep using the treatment, through a "free drug program."
The news follows data from a clinical trial of 664 ALS patients announced in March, which found no significant differences in outcomes between those on the treatment group and those who received a placebo.
It was a big blow for patients with amyotrophic lateral sclerosis, sometimes called Lou Gehrig's disease after the famous baseball player, which devastates nerve cells in the brain and spinal cord.
ALS affects about two people per 100,000 every year, causing progressive loss of motor and cognitive function. Most patients die within five years of their diagnosis.
Relyvrio's approval by the US Food and Drug Administration in 2022 was controversial and based on the results of a single trial that involved just 137 participants.
The FDA itself noted there was "residual uncertainty about the evidence of effectiveness" -- but "given the serious and life-threatening nature of ALS and the substantial unmet need, this level of uncertainty is acceptable in this instance and consideration of these results in the context of regulatory flexibility is appropriate."
- Patient groups backed approval -
Advocacy groups also mounted a major campaign sending a petition to the FDA with tens of thousands of signatures urging approval. Once it became available, Amylyx reportedly announced an eye-watering list price of $158,000 per year in the US, drawing criticism.
Patient groups in Europe watched with desperation at the bureaucratic delays.
When the European Union drug watchdog later announced it was rejecting Relyvrio, the decision was slammed as "an affront" by angry French patients, who say they "don't have time to wait." France later relented, offering conditional approval in November.
"We commend Amylyx for pulling Relyvrio off the market, while still ensuring that people living with ALS can access the drug if they believe it is helping them," said the US-based ALS association, which had lobbied for the drug's approval and funded its research.
"Safe and potentially effective treatments can be made accessible rapidly until further research can confirm their efficacy," it added.
For now, there remain only a handful of treatments available.
Riluzole, FDA approved in 1995, prolongs life approximately three months. Edaravone, FDA approved in 2017, has been found to slow disease progression and improve survival.
And in 2023, the regulatory body approved tofersen, a gene therapy treatment that targets those ALS cases that are caused by mutations in the SOD1 gene.
K.Thomson--BTB



