
-
 UK foreign minister stresses 'urgent need' to reopen Hormuz strait
UK foreign minister stresses 'urgent need' to reopen Hormuz strait
-
Macron says Trump marriage jibe does not 'merit response'

-
 Russia will send second ship with oil to Cuba: minister
Russia will send second ship with oil to Cuba: minister
-
Belgian bishop takes on Vatican with push to ordain married men

-
 Oil rallies, stocks drop as Trump dampens Mideast hopes
Oil rallies, stocks drop as Trump dampens Mideast hopes
-
Nexperia's China unit nears fully local production of chips: company sources

-
 Indonesia issues fresh summons for Google, Meta over teen social media ban
Indonesia issues fresh summons for Google, Meta over teen social media ban
-
Japan axe coach Nielsen 12 days after winning Women's Asian Cup

-
 French President Macron lands in South Korea after Japan visit
French President Macron lands in South Korea after Japan visit
-
India's says defence exports hit 'all-time high' of $4 bn

-
 Nielsen leaves as Japan coach weeks after winning Women's Asian Cup
Nielsen leaves as Japan coach weeks after winning Women's Asian Cup
-
Too bright: Seoul to dim digital billboards after complaints

-
 Iran vows 'crushing' attacks on US after Trump threats
Iran vows 'crushing' attacks on US after Trump threats
-
Women's Asian Cup finalists accuse governing body over equal money

-
 French president Macron heads to South Korea after Japan visit
French president Macron heads to South Korea after Japan visit
-
Armenia's underground salt clinic at centre of alternative medicine debate

-
 'Muted' international response as Senegal enacts same-sex relations law
'Muted' international response as Senegal enacts same-sex relations law
-
Slow boat to Ilulissat: long nights on Greenland's last ferry

-
 Wemby rampant again as Spurs rack up 10th straight win
Wemby rampant again as Spurs rack up 10th straight win
-
Ukrainian death metal band growls against Russia's war

-
 Iran fires missiles at Israel after Trump threatens weeks of strikes
Iran fires missiles at Israel after Trump threatens weeks of strikes
-
Surging 'Jewish terrorism' in West Bank condemned but unpunished

-
 England's Brook, Bethell warned after New Zealand nightclub incident
England's Brook, Bethell warned after New Zealand nightclub incident
-
What's real anymore? AI warps truth of Middle East war

-
 Europe to negotiate with NASA on lunar missions: ESA
Europe to negotiate with NASA on lunar missions: ESA
-
Trump tells US that Iran war victory near, but vows big strikes

-
 Poppies offer hope in fire-scarred Los Angeles
Poppies offer hope in fire-scarred Los Angeles
-
Trump says Iran war almost over, warns of weeks more heavy strikes

-
 Oil rallies, stocks tumble as Trump says US to hammer Iran further
Oil rallies, stocks tumble as Trump says US to hammer Iran further
-
US Republicans announce deal to end partial government shutdown

-
 Trump tells Americans that Iran war ending as popularity dips
Trump tells Americans that Iran war ending as popularity dips
-
7.4-magnitude quake off Indonesia kills one, tsunami warning lifted

-
 Bordeaux-Begles' Van Rensburg 'not thinking' about Champions Cup double
Bordeaux-Begles' Van Rensburg 'not thinking' about Champions Cup double
-
Side-by-Side Conference Hosts Close to 800 Participants in Malta to Advance Women's Health Care

-
 U.S. Polo Assn. Supports Division I National Intercollegiate Championship, Showcasing the Future of the Sport of Polo
U.S. Polo Assn. Supports Division I National Intercollegiate Championship, Showcasing the Future of the Sport of Polo
-
US automakers report mixed sales as car market awaits war impact

-
 Astronauts begin NASA lunar mission after climactic blast-off
Astronauts begin NASA lunar mission after climactic blast-off
-
Astronauts blast off for historic US lunar journey

-
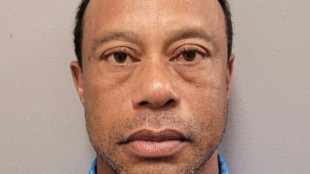
 Embattled Woods won't captain 2027 Ryder Cup team: PGA of America
Embattled Woods won't captain 2027 Ryder Cup team: PGA of America
-
Judge allows Woods to travel overseas for treatment
-
 Chelsea's Bompastor furious as Arsenal reach women's Champions League semis
Chelsea's Bompastor furious as Arsenal reach women's Champions League semis
-
US lifts sanctions on Venezuelan interim leader Delcy Rodriguez

-
 Arsenal resist Chelsea rally to reach women's Champions League semis
Arsenal resist Chelsea rally to reach women's Champions League semis
-
Defending champ Pegula wins WTA Charleston opener

-
 New frog species carrying eggs on back discovered in Peru
New frog species carrying eggs on back discovered in Peru
-
Benfica winger Prestianni denies 'ugly' racism claims

-
 Tuchel casts doubt on Foden's World Cup chances
Tuchel casts doubt on Foden's World Cup chances
-
Slot hoping Salah can still burnish Liverpool legacy

-
 Astronauts strapped in for historic US lunar launch
Astronauts strapped in for historic US lunar launch
-
Top World Bank official 'extremely concerned' by fallout of Iran war


European medicines watchdog rejects new Alzheimer's drug
Europe's medicines watchdog on Friday rejected a marketing request for a new Alzheimer's disease treatment, saying the risks of the medicine's side effects, including potential brain bleeding, outweighed the benefits.
The decision by the Amsterdam-based European Medicines Agency was met with dismay, but experts said effective treatment for the degenerative mental disease affecting millions in Europe alone, was getting closer.
"The CHMP recommended not granting a marketing authorisation for Leqembi, a medicine intended for the treatment of Alzheimer's disease," the European Medicines Agency said, referring to its committee for evaluating drugs for human use.
Leqembi, which was developed by US multinational Biogen and Japanese-based Eisai, is the brand name of an active substance called lecanemab, which is used to treat adults with mild memory and cognitive problems resulting from the early stages of the common type of dementia.
But the CHMP said "the observed effect of Leqembi on delaying cognitive decline does not counterbalance the risk of serious side events associated with the medicine."
"The most important safety concern with Leqembi is the frequent occurrence of amyloid-related imaging abnormalities (ARIA), a side effect, seen in brain imaging, that involves swelling and potential bleedings in the brain," the EMA said.
- 'Unmet need' -
Leqembi is a monoclonal antibody, a type of protein that clings to a substance in the brain and can delay worsening of the disease. It is given intravenously every two weeks.
Leqembi, together with another Alzheimer's drug called Aduhelm -- also developed by Biogen and Eisai -- received approval from the US Food and Drug Administration (FDA) early last year.
Both drugs were approved through an accelerated process by the FDA for drugs treating serious conditions where there is an unmet medical need.
But in late January this year, Biogen pulled the controversial Aduhelm from the market, saying it was focusing on Leqembi instead.
Preliminary data from a trial of Leqembi was released in September 2022 and found it slowed cognitive decline in Alzheimer's patients by 27 percent.
Around eight million people in the European Union live with dementia, with Alzheimer's disease accounting for more than half of these cases, according to the Alzheimer Europe website.
Eisai, in a statement, said it was "extremely disappointed with the CHMP's negative opinion".
"There is a significant unmet need for new innovative treatment options that target an underlying cause of disease progression," Eisai's chief clinical officer Lynn Kramer said.
Eisai said it would seek a re-examination of the EMA's opinion "to ensure this treatment is available for eligible people living with early Alzheimer's disease in the EU as soon as possible."
The EMA said Eisai presented a main study involving 1,795 people with early Alzheimer's who either received Leqembi or a placebo, measured over a span of 18 months.
Eisai said that the watchdog added that its refusal had "no consequences for patients in clinical trials with Leqembi".
Eisai may now ask for a re-examination within 15 days, the EMA said.
- 'Ramp up efforts' -
Experts voiced disappointment at the EMA's refusal, but added there were "reasons to remain hopeful".
"Lecanemab has shown that it is possible to slow down disease progression, and research does work," said Tara Spires-Jones, president of the British Neuroscience Association.
"Now we need to ramp up our efforts to discover new and safer treatments," she said in a statement, adding that "each discovery brings us closer to new and better treatments."
Bart De Strooper, a professor in Alzheimer's disease at the University College London called the EMA's decision "unfortunate yet not unexpected".
"This conservative approach means that patients and doctors eager to explore a proven effective drug are now denied access," he said in a statement.
"With no current therapies available, it's disheartening to think that if we had applied such caution in the past, particularly with cancer drugs and their severe side effects, we might still be without cancer treatments today," De Strooper said.
C.Kovalenko--BTB



