
-
 UK foreign minister stresses 'urgent need' to reopen Hormuz strait
UK foreign minister stresses 'urgent need' to reopen Hormuz strait
-
Macron says Trump marriage jibe does not 'merit response'

-
 Russia will send second ship with oil to Cuba: minister
Russia will send second ship with oil to Cuba: minister
-
Belgian bishop takes on Vatican with push to ordain married men

-
 Oil rallies, stocks drop as Trump dampens Mideast hopes
Oil rallies, stocks drop as Trump dampens Mideast hopes
-
Nexperia's China unit nears fully local production of chips: company sources

-
 Indonesia issues fresh summons for Google, Meta over teen social media ban
Indonesia issues fresh summons for Google, Meta over teen social media ban
-
Japan axe coach Nielsen 12 days after winning Women's Asian Cup

-
 French President Macron lands in South Korea after Japan visit
French President Macron lands in South Korea after Japan visit
-
India's says defence exports hit 'all-time high' of $4 bn

-
 Nielsen leaves as Japan coach weeks after winning Women's Asian Cup
Nielsen leaves as Japan coach weeks after winning Women's Asian Cup
-
Too bright: Seoul to dim digital billboards after complaints

-
 Iran vows 'crushing' attacks on US after Trump threats
Iran vows 'crushing' attacks on US after Trump threats
-
Women's Asian Cup finalists accuse governing body over equal money

-
 French president Macron heads to South Korea after Japan visit
French president Macron heads to South Korea after Japan visit
-
Armenia's underground salt clinic at centre of alternative medicine debate

-
 'Muted' international response as Senegal enacts same-sex relations law
'Muted' international response as Senegal enacts same-sex relations law
-
Slow boat to Ilulissat: long nights on Greenland's last ferry

-
 Wemby rampant again as Spurs rack up 10th straight win
Wemby rampant again as Spurs rack up 10th straight win
-
Ukrainian death metal band growls against Russia's war

-
 Iran fires missiles at Israel after Trump threatens weeks of strikes
Iran fires missiles at Israel after Trump threatens weeks of strikes
-
Surging 'Jewish terrorism' in West Bank condemned but unpunished

-
 England's Brook, Bethell warned after New Zealand nightclub incident
England's Brook, Bethell warned after New Zealand nightclub incident
-
What's real anymore? AI warps truth of Middle East war

-
 Europe to negotiate with NASA on lunar missions: ESA
Europe to negotiate with NASA on lunar missions: ESA
-
Trump tells US that Iran war victory near, but vows big strikes

-
 Poppies offer hope in fire-scarred Los Angeles
Poppies offer hope in fire-scarred Los Angeles
-
Trump says Iran war almost over, warns of weeks more heavy strikes

-
 Oil rallies, stocks tumble as Trump says US to hammer Iran further
Oil rallies, stocks tumble as Trump says US to hammer Iran further
-
US Republicans announce deal to end partial government shutdown

-
 Trump tells Americans that Iran war ending as popularity dips
Trump tells Americans that Iran war ending as popularity dips
-
7.4-magnitude quake off Indonesia kills one, tsunami warning lifted

-
 Bordeaux-Begles' Van Rensburg 'not thinking' about Champions Cup double
Bordeaux-Begles' Van Rensburg 'not thinking' about Champions Cup double
-
Side-by-Side Conference Hosts Close to 800 Participants in Malta to Advance Women's Health Care

-
 U.S. Polo Assn. Supports Division I National Intercollegiate Championship, Showcasing the Future of the Sport of Polo
U.S. Polo Assn. Supports Division I National Intercollegiate Championship, Showcasing the Future of the Sport of Polo
-
US automakers report mixed sales as car market awaits war impact

-
 Astronauts begin NASA lunar mission after climactic blast-off
Astronauts begin NASA lunar mission after climactic blast-off
-
Astronauts blast off for historic US lunar journey

-
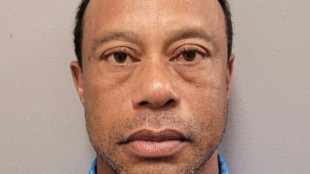
 Embattled Woods won't captain 2027 Ryder Cup team: PGA of America
Embattled Woods won't captain 2027 Ryder Cup team: PGA of America
-
Judge allows Woods to travel overseas for treatment
-
 Chelsea's Bompastor furious as Arsenal reach women's Champions League semis
Chelsea's Bompastor furious as Arsenal reach women's Champions League semis
-
US lifts sanctions on Venezuelan interim leader Delcy Rodriguez

-
 Arsenal resist Chelsea rally to reach women's Champions League semis
Arsenal resist Chelsea rally to reach women's Champions League semis
-
Defending champ Pegula wins WTA Charleston opener

-
 New frog species carrying eggs on back discovered in Peru
New frog species carrying eggs on back discovered in Peru
-
Benfica winger Prestianni denies 'ugly' racism claims

-
 Tuchel casts doubt on Foden's World Cup chances
Tuchel casts doubt on Foden's World Cup chances
-
Slot hoping Salah can still burnish Liverpool legacy

-
 Astronauts strapped in for historic US lunar launch
Astronauts strapped in for historic US lunar launch
-
Top World Bank official 'extremely concerned' by fallout of Iran war


US health regulator rejects MDMA treatment for PTSD, for now
US health regulators on Friday denied an application for treating post-traumatic stress disorder (PTSD) with the drug MDMA, commonly known as ecstasy, saying more investigation needed to be done.
The company that submitted the application, Lykos Therapeutics, said in a statement that the Food and Drug Administration (FDA) had requested an additional Phase 3 clinical trial to study MDMA's "safety and efficacy."
A panel of experts convened by the FDA to evaluate clinical data on MDMA had overwhelmingly voted in early June to say there was insufficient evidence to prove it was effective.
While unsurprising, the decision announced Friday represents a blow to advocates of the novel treatment.
"The FDA request for another study is deeply disappointing, not just for all those who dedicated their lives to this pioneering effort, but principally for the millions of Americans with PTSD... who have not seen any new treatment options in over two decades, said Lykos CEO Amy Emerson.
PTSD is a debilitating mental health condition that develops after a person experiences or is threatened by traumatic events such as death, combat or sexual assault.
It affects an estimated five percent of Americans in any given year.
Pharmaceutical treatment options for PTSD are so far limited to two antidepressants that require three months of dosing to take effect, and response rates to the medications have been found to be uneven.
MDMA -- methylenedioxymethamphetamine -- is a Schedule 1 drug under the Controlled Substances Act, and approving it for medical use would have represented a major shift.
California-based Lykos based its request for regulatory approval on two clinical studies, each of which enrolled around 100 people, to evaluate MDMA used together with other psychological interventions such as talk therapy, against a placebo with talk therapy.
These two studies, published in the prestigious journal Nature Medicine, indicated MDMA was indeed both safe and highly effective at treating PTSD.
But nine out of 11 experts on the FDA panel said available data was not enough to show the treatment was effective, and 10 out of 11 said the benefits did not outweigh the risks.
In a briefing document put together ahead of the meeting, FDA staff raised concerns about Lykos's clinical trial methodology and criticized the company for not gathering sufficient side effect data.
The company said it will "work diligently in the coming months to address FDA's concerns and to take advantage of agency processes to resolve scientific disagreements."
"We intend to work tirelessly and use all available regulatory pathways to find a reasonable and expeditious path forward," Emerson added.
C.Meier--BTB



